
Electron Configuration For Manganese Atomic Number 25 How Do You Draw
Electron configurations are the summary of where the electrons are around a nucleus. As we learned earlier, each neutral atom has a number of electrons equal to its number of protons.
Sharing the Teachings Without Name and Number Urantia Book Urantia
Electron Configurations are useful for: Determining the valency of an element. Predicting the properties of a group of elements (elements with similar electron configurations tend to exhibit similar properties). Interpreting atomic spectra.
M7Q7 Electron Configurations, Orbital Box Notation Chem 103/104
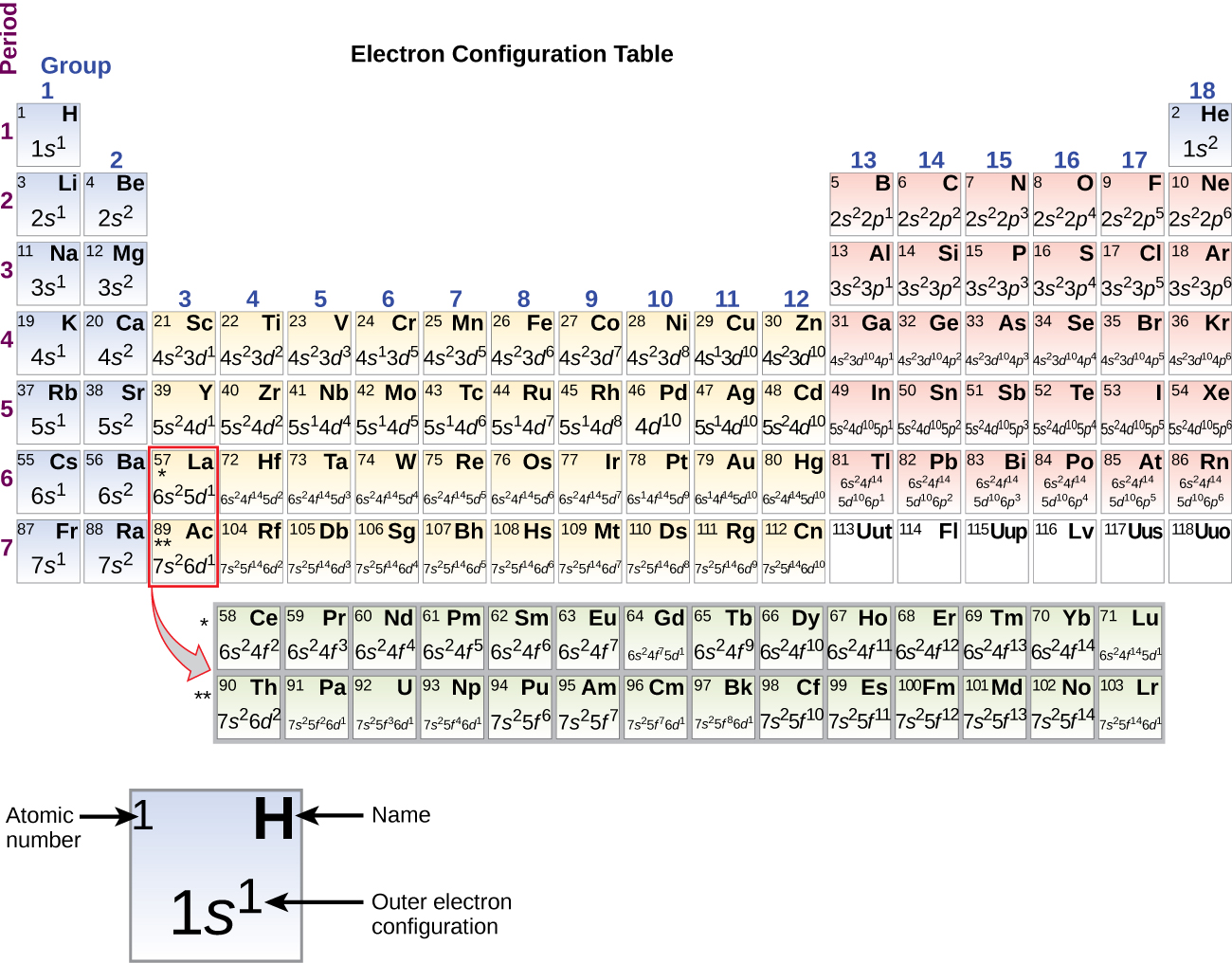
The third major category of elements arises when the distinguishing electron occupies an f subshell. The first example occurs in the case of the lanthanoids (elements having atomic numbers between 57 and 71).The lanthanoids have the general electron configuration [Kr]4d 10 4f i 5s 2 5p 6 5d 0 or 1 6s 2. where i is a number between 0 and 14. Thus in the building-up process for the lanthanoids.
/800px-Orbital_representation_diagram.svg-589bd6285f9b58819cfd8460.png)
Electron Configuration Chart
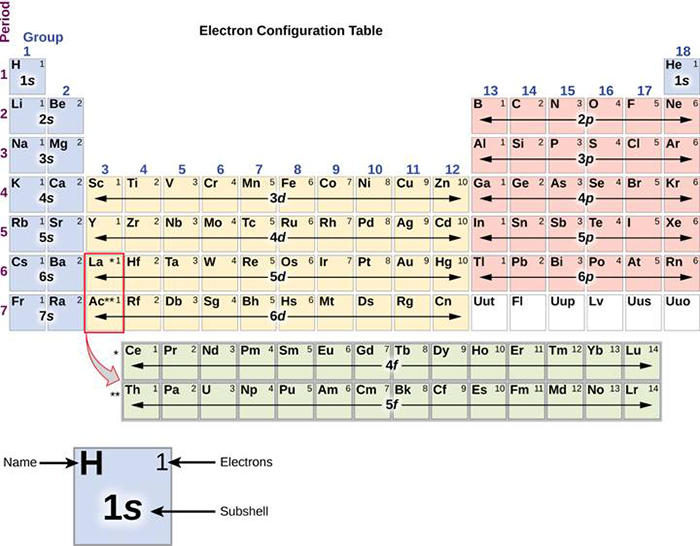
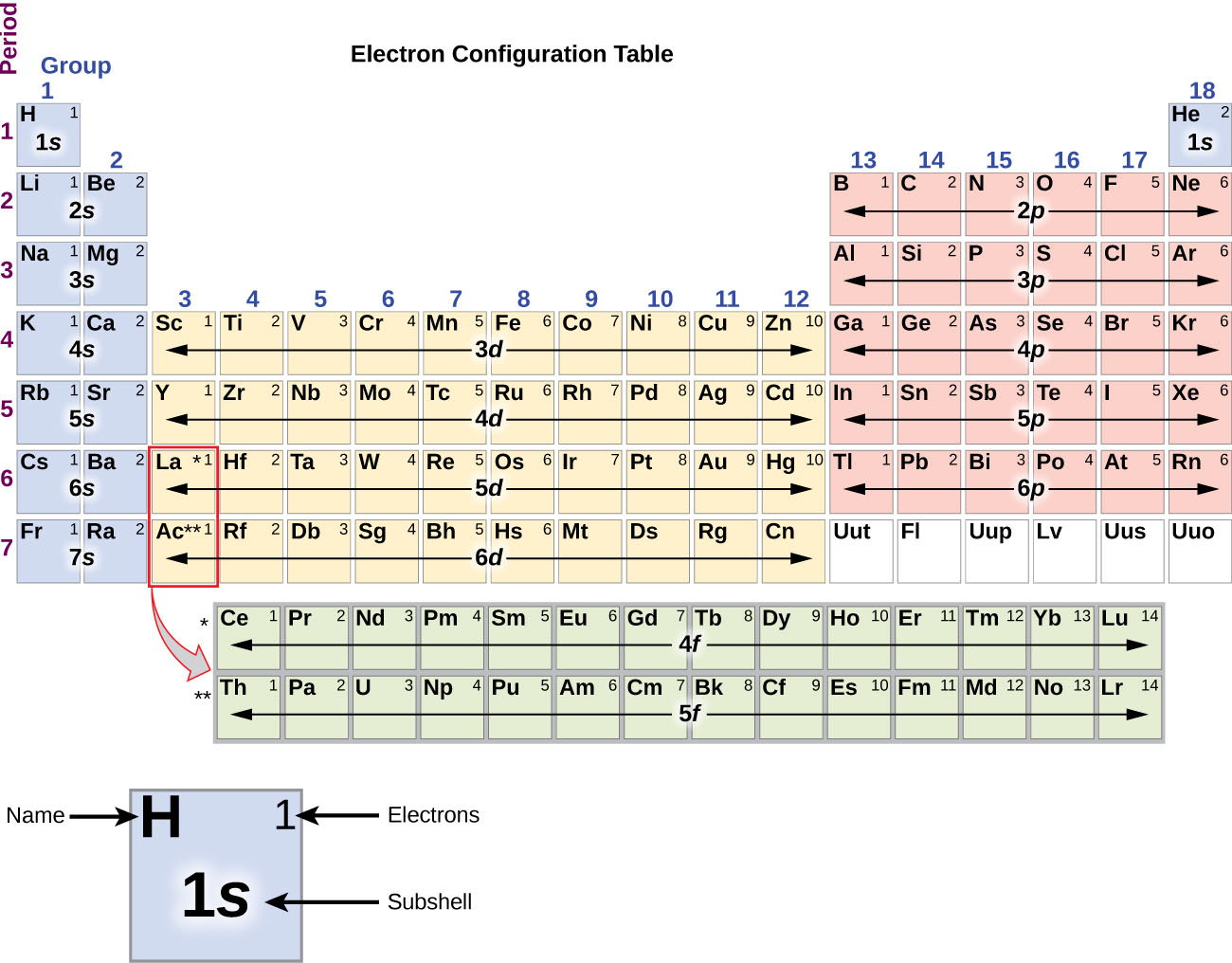
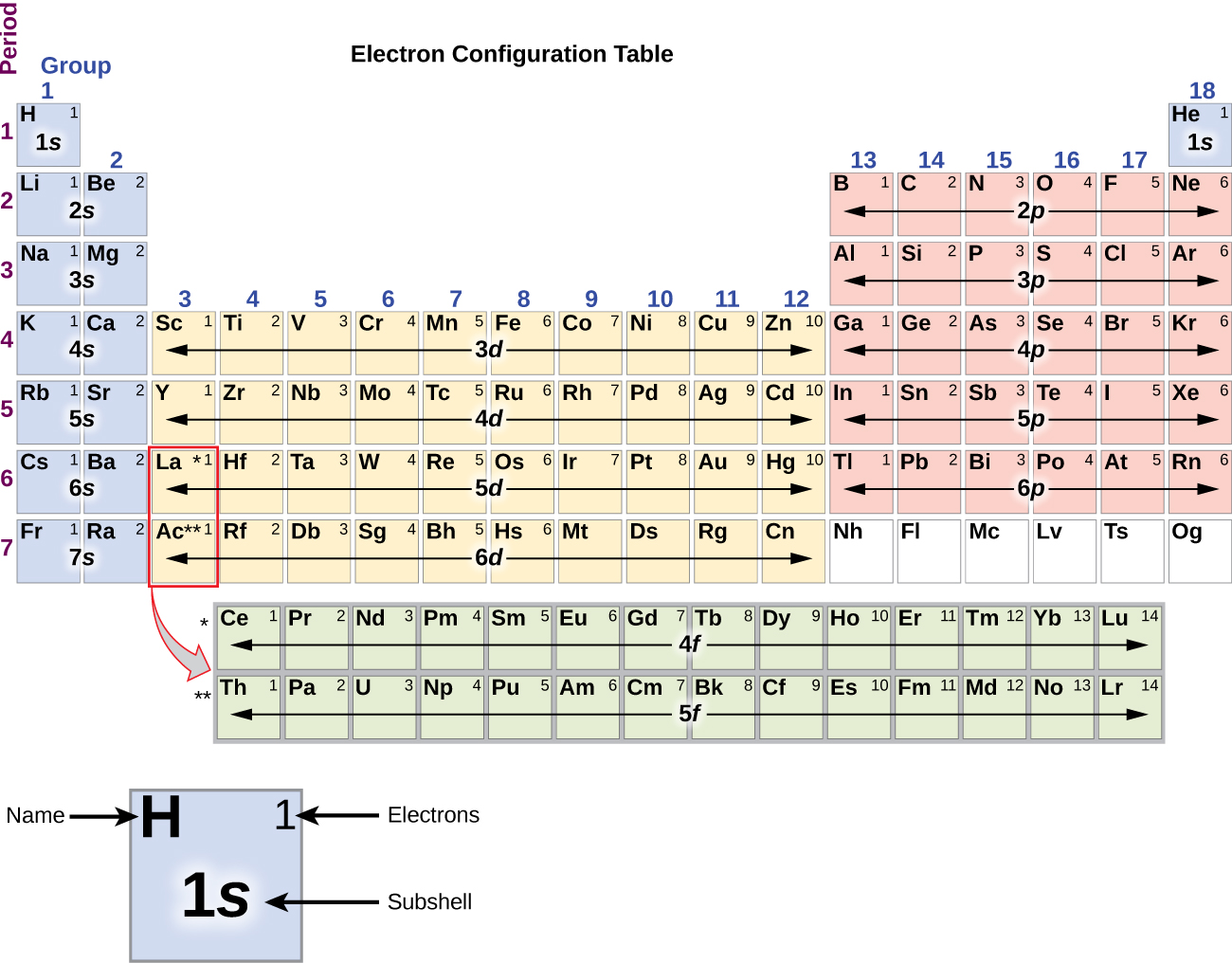
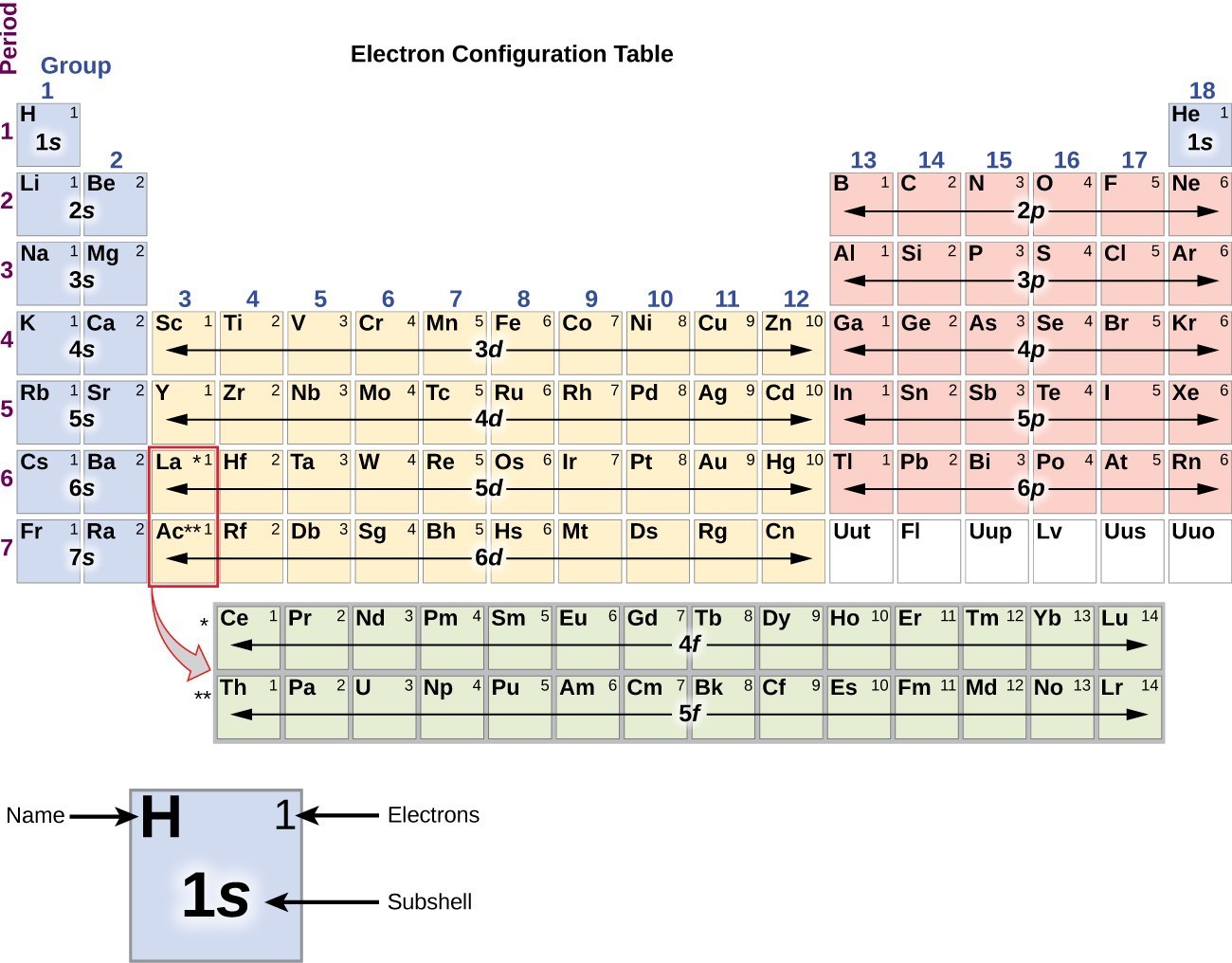
The arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom. We describe an electron configuration with a symbol that contains three pieces of information ( Figure 6.25 ): The number of the principal quantum shell, n,
Question 9267e Socratic
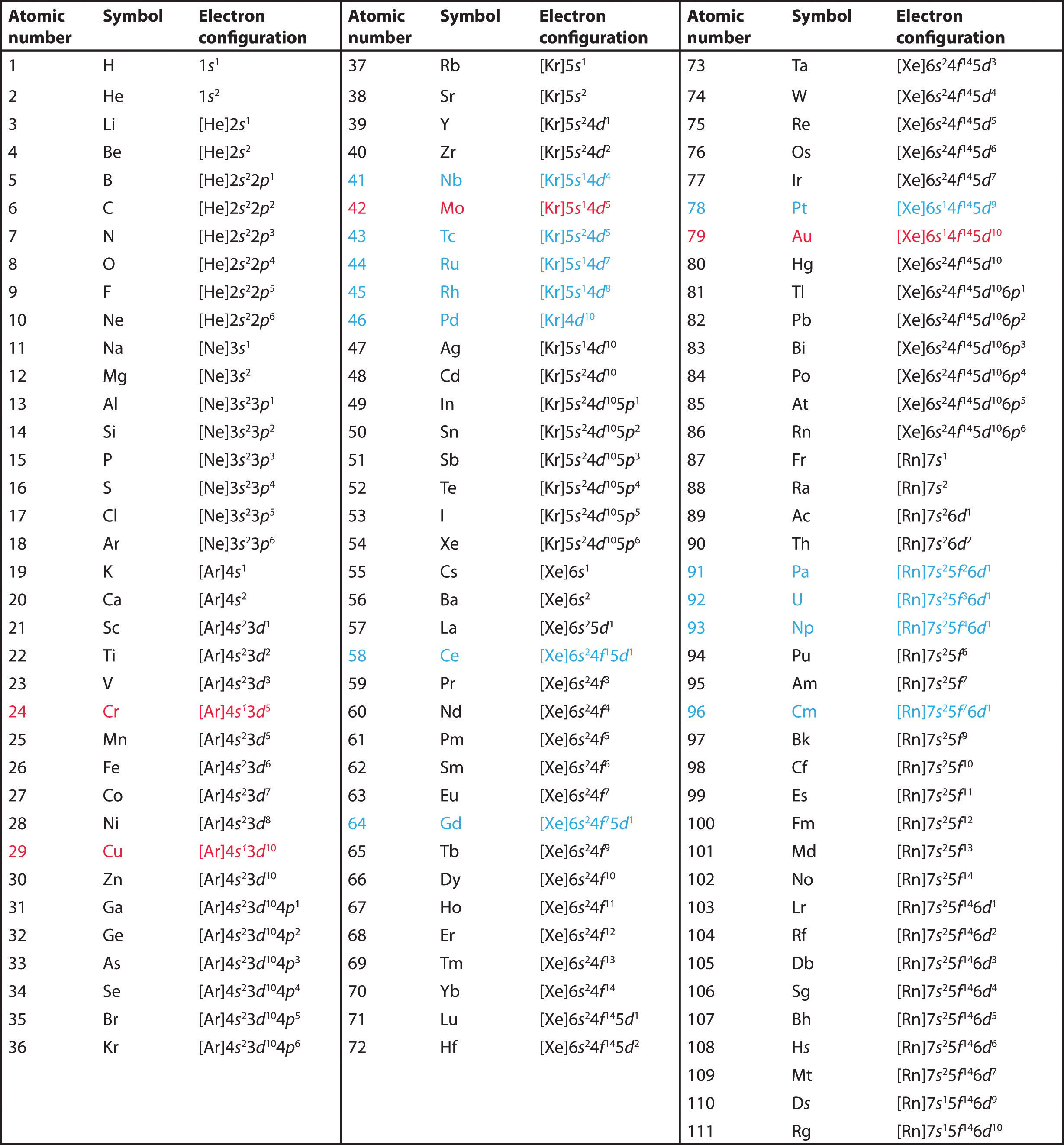
The same rule will apply to transition metals when forming ions. You should note that the ns electrons are always lost before the (n-1)d when forming cations for transition metals.For example, the electron configuration for Zn: [Ar]4s 2 3d 10 . the electron configuration for Zn +2: [Ar]3d 10 . The transition metals still do not end up being isoelectronic with a noble gas, but the loss of two.
Nitrogen Element With Reaction, Properties, Uses, & Price Periodic Table
Electron Configuration Notation: -shows the arrangment of electrons around the nucleus of an atom. - helps chemist understanding how elements form chemical bonds. - can be written using the period table or an electron configuration chart. How to Write the Electron Configuration for Nitrogen (N)
Predict the Ground State Electron Configuration Co3+ Denker Ensterly
The arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom. We describe an electron configuration with a symbol that contains three pieces of information ( Figure 3.1.2 3.1. 2 ): The number of the principal quantum shell, n, The letter that designates the orbital type (the subshell, l ), and

Electron Configuration Chart Electron configuration, Atomic structure
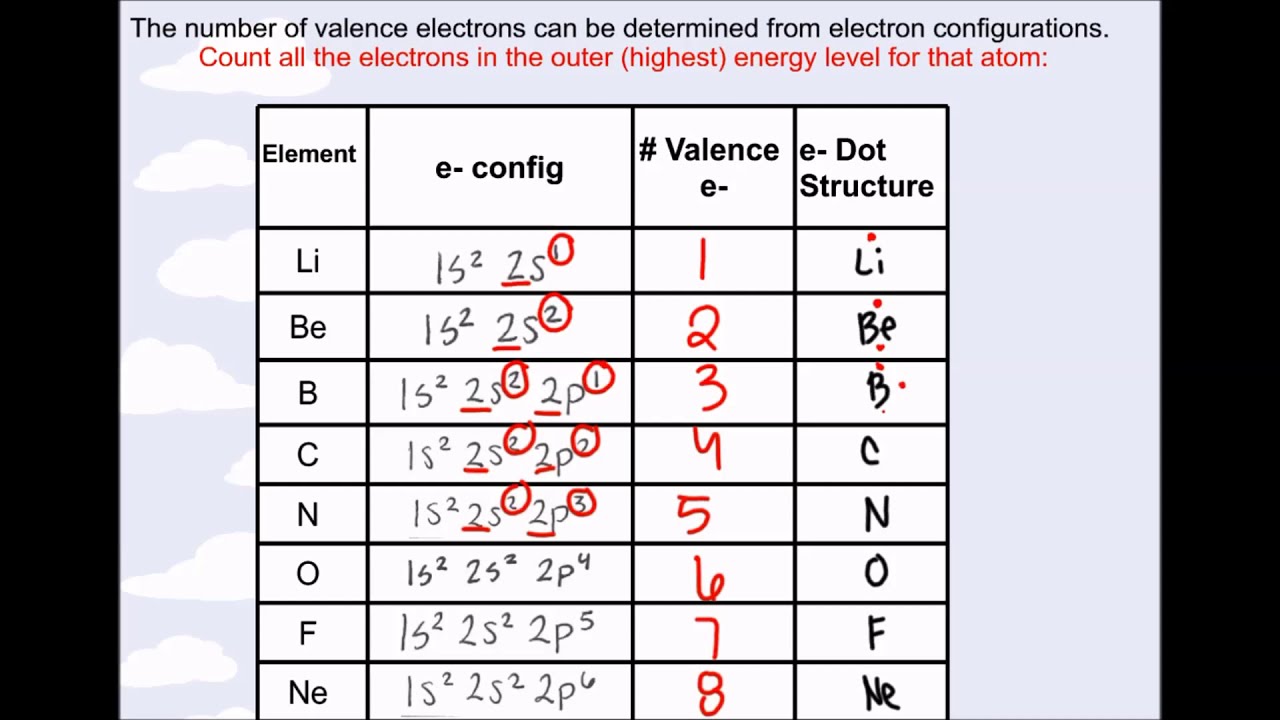
Electron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table.. The electron configuration for the first 10 elements. H #1s^1# He #1s^2# Li #1s^2 2s^1# Be #1s^2 2s^2# B #1s^2 2s^2 2p^1# C #1s^2 2s^2 2p^2# N #1s^2 2s^2 2p^3# O #1s^2 2s^2 2p^4# F #1s^2 2s^2 2p^5#
7.8B Electron Configurations and the Periodic Table Chemistry LibreTexts
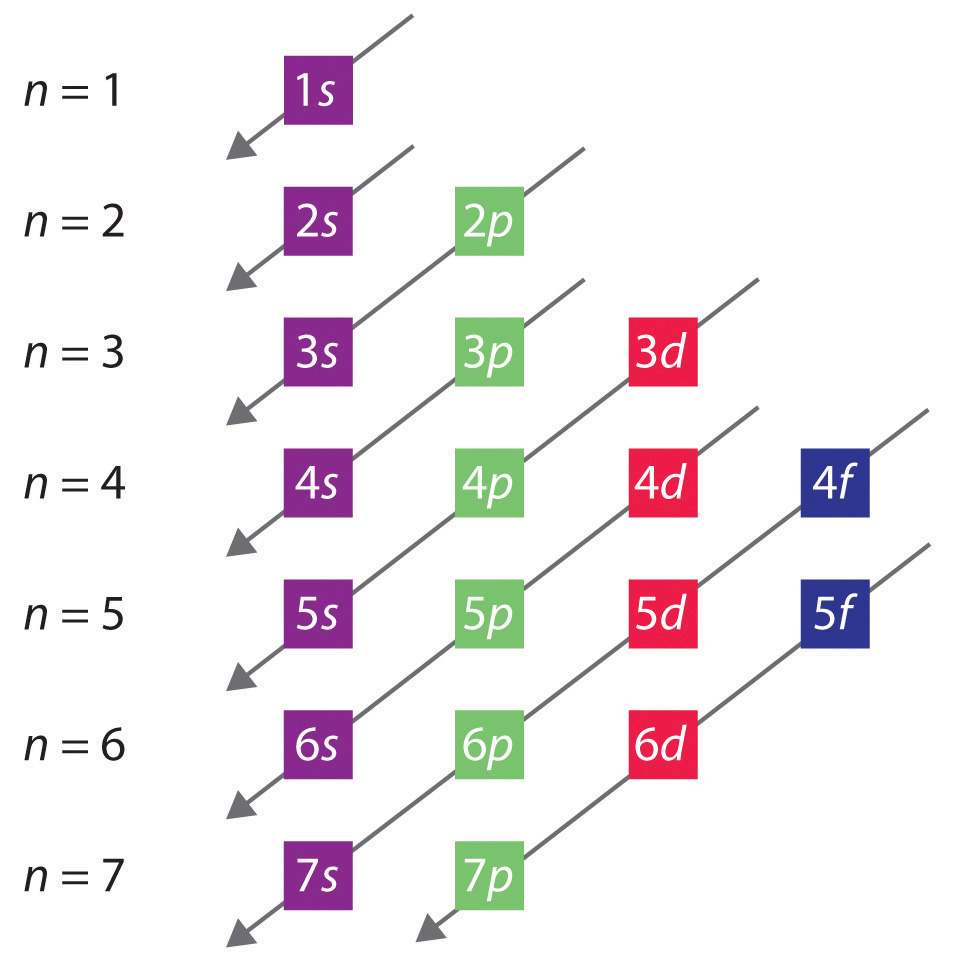
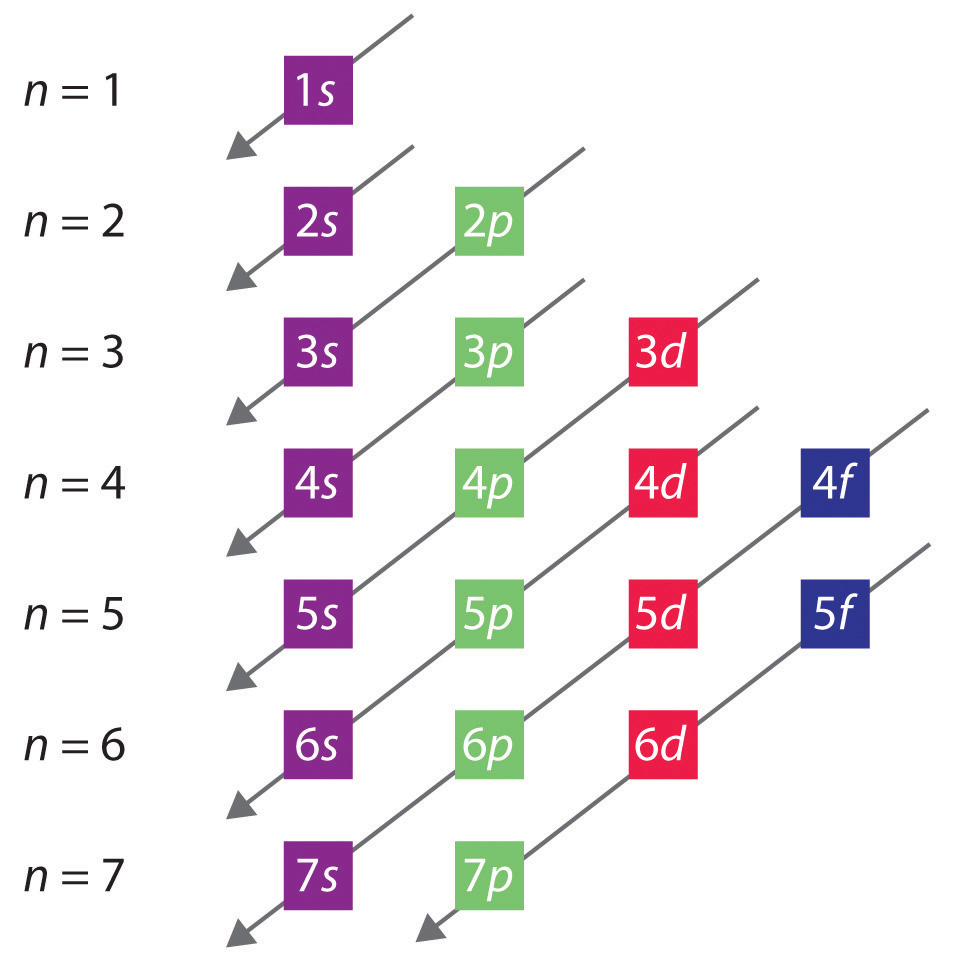
The numbers of electrons that can occupy each shell and each subshell arise from the equations of quantum mechanics, [a] in particular the Pauli exclusion principle, which states that no two electrons in the same atom can have the same values of the four [2]
Valence Electrons & Electron Configurations YouTube
Electron configurations are a simple way of writing down the locations of all of the electrons in an atom. As we know, the positively-charged protons in the nucleus of an atom tend to attract negatively-charged electrons.
Electronic Structure of Atoms (Electron Configurations) Chemistry
In this case, 2+2+6+2+6+2+10+6+2+1= 39 and Z=39, so the answer is correct. A slightly more complicated example is the electron configuration of bismuth (symbolized Bi, with Z = 83). The periodic table gives the following electron configuration: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p65s2 4d10 5p6 6s2 4f14 5d10 6p3.
Electron Configurations, Orbital Box Notation (M7Q7) UWMadison
Principal Quantum Number (n) The principal quantum number n indicates the shell or energy level in which the electron is found. The value of n can be set between 1 to n, where n is the value of the outermost shell containing an electron. This quantum number can only be positive, non-zero, and integer values. That is, n=1,2,3,4,.. For example, an Iodine atom has its outmost electrons in the 5p.

Electron Configurations YouTube
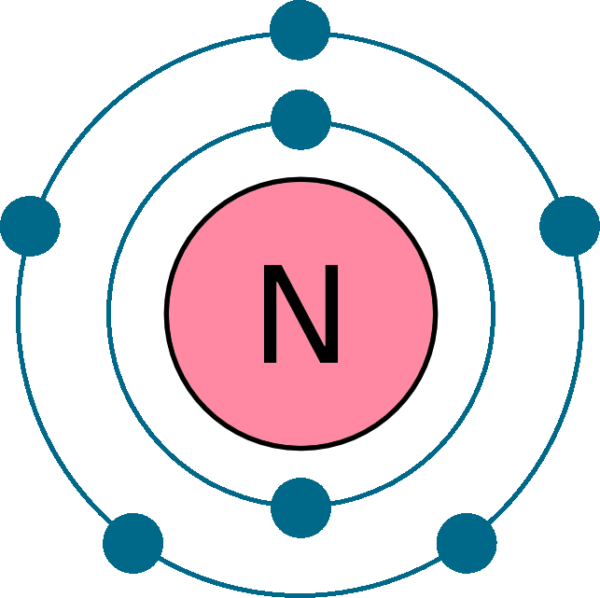
The arrangement of electrons in nitrogen in specific rules in different orbits and orbitals is called the electron configuration of nitrogen. The electron configuration of nitrogen is [ He] 2s 2 2p 3 , if the electron arrangement is through orbitals. Electron configuration can be done in two ways.

Electronic Configurations Intro Chemistry LibreTexts
1). You can effortlessly find every single detail about the elements from this single Interactive Periodic table. 2). You will get the detailed information about the periodic table which will convert a newbie into pro. 3). You will also get the HD images of the Periodic table (for FREE).
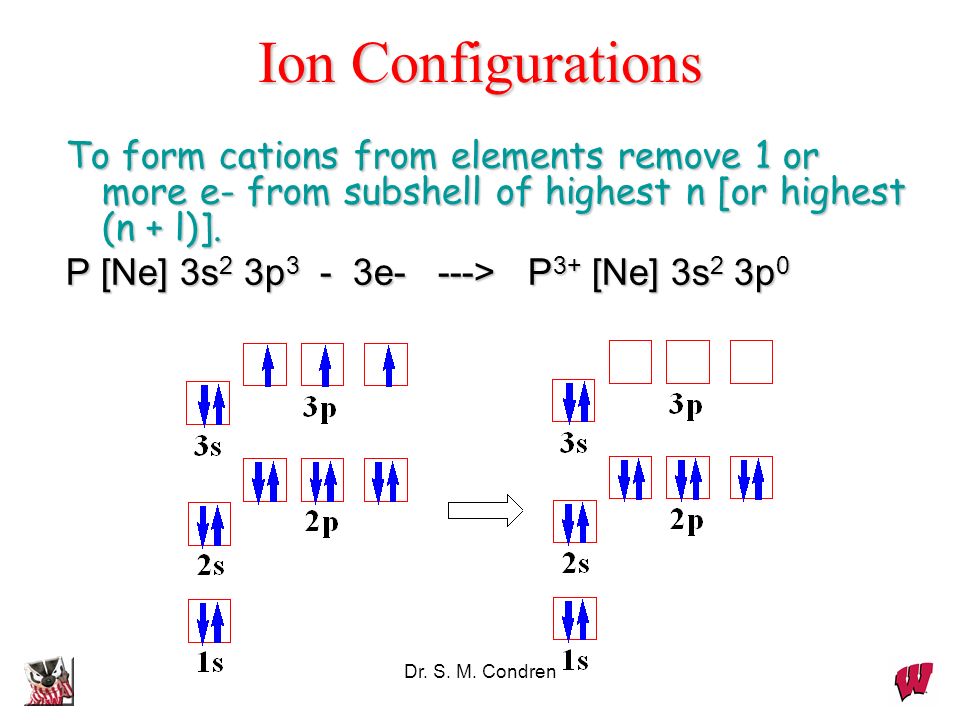
P [Ne] 3s2 3p3 3e —> P3+ [Ne] 3s2 3p0. Dr. S. M. Condren. Dynamic
This electron configuration calculator will instantly show you the distribution of electrons in the orbitals of any periodic element you choose. Typically, you need at least 8 steps to determine the electron configuration, starting with finding the atomic number by looking at the list of orbitals and understanding the notation.
Use The Orbital Diagram For Nitrogen To Write Quantum Numbers For The
AboutTranscript. Electron configurations describe where electrons are located around the nucleus of an atom. For example, the electron configuration of lithium, 1s²2s¹, tells us that lithium has two electrons in the 1s subshell and one electron in the 2s subshell. Created by Sal Khan.